Stereoselectivity of four (R)-selective transaminases for the asymmetric amination of ketones
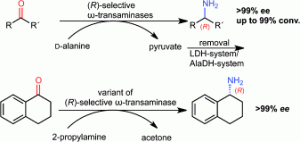
Four (R)-ω-transaminases originating from Hyphomonas neptunium (HN-ωTA), Aspergillus terreus (AT-ωTA) and Arthrobacter sp. (ArR-ωTA), as well as an evolved transaminase (ArRmut11-ωTA) were successfully employed for the amination of prochiral ketones leading to optically pure (R)-amines. The first three transaminases displayed perfect stereoselectivity for the amination of all substrates tested (ee >99%). Furthermore, the transaminase AT-ωTA led in most cases to better conversion than ArR-ωTA and HN-ωTA using D-alanine as amine donor. α-Tetralone, which was the only substrate not accepted by HN-ωTA, ArR-ωTA, and AT-ωTA, was successfully transformed with perfect enantioselectivity (ee >99%) into the corresponding optically pure amine employing the variant ArRmut11-ωTA.


