Mechanistic Insight into the Catalytic Promiscuity of Amine Dehydrogenases: Asymmetric Synthesis of Secondary and Primary Amines
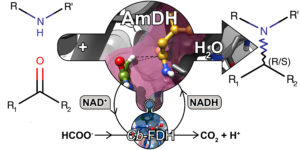
Biocatalytic asymmetric aminations of ketones using amine dehydrogenases (AmDHs) or transaminases, are efficient methods for the synthesis of α‐chiral primary amines. A major challenge is the extension of the amination to the synthesis of secondary and tertiary amines. Herein, we show for the first time that AmDHs are capable of accepting other amine donors, hence giving access to enantioenriched secondary amines with conversions up to 43%. Surprisingly, in several cases, we observed the promiscuous formation of enantiopure primary amines along with the expected secondary amines. By conducting practical laboratory experiments and computational experiments, we propose that the promiscuous formation of primary amines along with secondary amines is due to an unprecedented nicotinamide (NAD)‐dependent formal transamination catalysed by AmDHs. In nature, this type of mechanism is commonly performed by pyridoxal 5’‐phosphate (PLP) aminotransferase and not by dehydrogenases. Finally, we propose a catalytic pathway that rationalises the promiscuous NAD‐dependent formal transamination activity and explains the formation of the observed mixture of products. This work increases our understanding on the catalytic mechanism of NAD‐dependent aminating enzymes such as AmDHs and will aid further research on the rational engineering of oxidoreductases for the synthesis of α‐chiral secondary and tertiary amines.