Conversion of alcohols to enantiopure amines through dual enzyme hydrogen-borrowing cascades
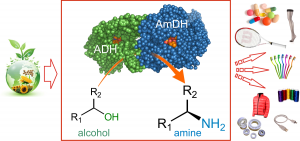
A biocatalytic hydrogen-borrowing amination of primary and secondary alcohols that allows for the efficient and environmentally benign production of enantiopure amines is presented. The method relies on a combination of two enzymes: an alcohol dehydrogenase operating in tandem with an amine dehydrogenase to aminate a structurally diverse range of aromatic and aliphatic alcohols, yielding up to 96% conversion and 99% enantiomeric excess. Primary alcohols were aminated with high conversion (up to 99%). This redox self-sufficient network possesses high atom efficiency, sourcing nitrogen from ammonium and generating water as the sole by-product.


