Asymmetric Preparation of prim-, sec-, and tert-amines employing selected biocatalysts
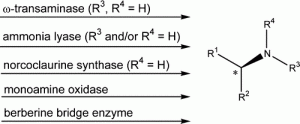
This account focuses on the application of ω-transaminases, lyases, and oxidases for the preparation of amines considering mainly work from our own lab. Examples are given to access α-chiral primary amines from the corresponding ketones as well as terminal amines from primary alcohols via a two-step biocascade. 2,6-Disubstituted piperidines, as examples for secondary amines, are prepared by biocatalytical regioselective asymmetric monoamination of designated diketones followed by spontaneous ring closure and a subsequent diastereoselective reduction step. Optically pure tert-amines such as berbines and N-methyl benzylisoquinolines are obtained by kinetic resolution via an enantioselective aerobic oxidative C–C bond formation.


