Asymmetric biocatalytic amination of ketones at the expense of ammonia and molecular hydrogen
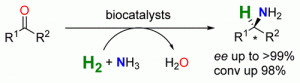
A biocatalytic system is presented for the stereoselective amination of ketones at the expense of NH3 and molecular hydrogen. By using a NAD+-reducing hydrogenase, an alanine dehydrogenase, and a suitable ω-transaminase, the R- as well as the S-enantiomer of various amines could be prepared with up to >99% ee and 98% conversion.


